Dimethyldichlorosilane (DMDCS/M2)
Product Name:Dimethyldichlorosilane (DMDCS)
Molecular Formula:(CH3)2SiCl2
Molecular Weight:129.1
CAS No.:75-78-5
UN No.:1162
Product Standard:GB/T 23953-2009 Physical and Chemical Properties:
Flash Point: -16℃(Closed cup) Melting Point: -76℃ Boiling Point: 71℃ Auto-ignition Temperature: 380℃ | Relative Density(water=1): 1.07 Relative Vapour density(air=1): 4.4 Vapour Pressure: 14.5 kPa (at 20℃) PH: Reacts with water to produce hydrogen chloride. |
Explosion Limits Lower[%(V/V)]: 3.4
Explosion Limits Upper[%(V/V)]: 9.5 Water Solubility: Reaction
Appearance: Colorless transparent liquid. with strong pungent smell
Structural formula: |
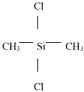
|
Technological Index: Item | Index |
Content of Dimethyldichlorosilane/% ≥ | 99.5 |
Content of Methyltrichlorosilane/% ≤ | 0.05 |
Properties and Uses
It is inflammable and easy to explode. It is a base material for manufacturing organic silicon intermediates, silicone oil, silicone rubber and silicone resins. It can also be used to manufacture super heat-resisting and high strong silicon carban fibe
Package, Storage and Handling
Steel drum/Platic drum,net weight 200kg/drum or ISO TANK. Storage at ventilate and dry place and prevention from water, heat and fire; It should be kept away from oxidant, acid and alkali. Handling according to hazardous substances and prevention from exposing to sunlight and rain. Be careful when loading and unloading to avoid damages ofthe package.
Chemical Stability: Stable in closed contain- ers under specified storage and handling conditions.
Conditions to Avoid: Incompatible materi- als, any sources of ignition or heat, exposure to moist air or water, electrical sparks.
Incompatibilities with Other Materials:
Strong oxidizing agents, water, alcohols, caustics, ammonia. Attacks many metals in presence of water. (Reacts violently with water to produce hydrogen chloride. Reacts violently with alcohols, amines causing fire and explosion hazard.)
Hazardous Decomposition Products: The substance decomposes on heating produc- ing toxic and corrosive fumes including hydrogen chloride, phosgene, and chlorine compounds. Reacts violently with water to produce hydrogen chloride.
 Product Information
Product Information
 Latest News
Latest News







